Notice that where the red is closest to Earth's surface is near large cities in the U.S.įind out more about TES and ozone on our greenhouse gas amazing fact page. In this image, the areas of highest ozone levels are shown in red. The profile shows with different colors how much ozone (or other greenhouse gas) is in the atmosphere at different altitudes. The TES mission is particularly interested in ozone and how it is distributed up and down throughout the atmosphere.Īs TES passes over Earth's surface, it gathers data that can be made into a profile of the atmosphere, like this one. hybrid orbitals, 432 Lewis structure, 399400 mole fraction in air at sea level. A spectrometer uses light to identify the chemical composition of matter. Different compounds having the same molecular formula are called isomers, and the prevalence of organic isomers reflects the extraordinary versatility of carbon. It's down here where we live-and pollute, unfortunately. The troposphere is the lowest part of the atmosphere. /rebates/&.com252fcarbon-dioxide-molecular-formula-image243393567. Carbon dioxide is made of molecules of a carbon atom bonded. This means that the chemical formula of water is HO. One instrument is called TES, which is short for Tropospheric Emission Spectrometer. In one molecule of water, there are two hydrogen atoms and one oxygen atom. They all study the atmosphere from about 20 miles high down to Earth's surface. n is equal to the number of atoms within the molecule of interest. The degrees of freedom for nonlinear molecules can be calculated using the formula: (2) 3 N 6. For example, carbon is used to reduce copper oxide to copper: Cu 2 O 1 C h 2Cu 1 CO Carbon monoxide is also used in metal processing it is used to reduce. NASA launched the Aura satellite in 2004 as part of its Earth Observing System. The degrees of vibrational modes for linear molecules can be calculated using the formula: (1) 3 N 5. Learning about Earth is part of NASA's mission. Vector illustration included icon amino acid, Molecule line icons. 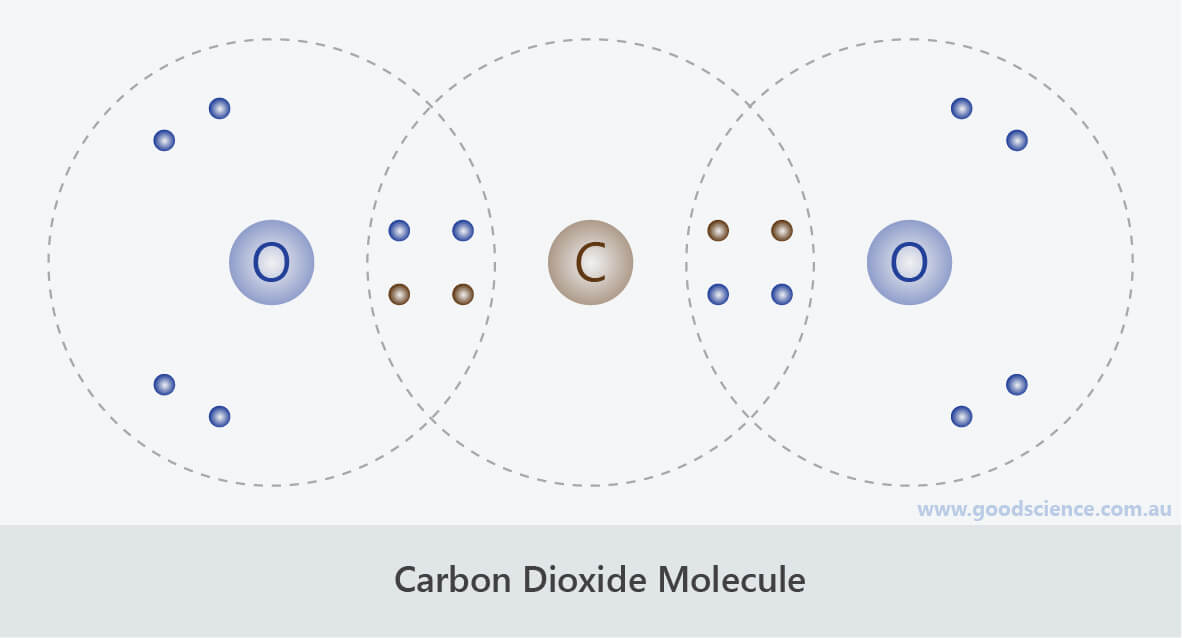
oxygen molecule global warming Sort by: Most popular Molecule line icons. Browse 610+ carbon dioxide chemical formula stock photos and images available, or start a new search to explore more stock photos and images. We must understand Earth in order to take good care of it. Browse 2,200+ carbon dioxide molecule stock photos and images available, or search for oxygen molecule or global warming to find more great stock photos and pictures. Free for commercial use High Quality Images. 78000+ Vectors, Stock Photos & PSD files. So, now that you know all your greenhouse gases, you might be wondering. Find & Download Free Graphic Resources for Carbon Dioxide Molecule. Now, take the big piece of construction paper and your crayons or felt pen and label it something like this: You will need only one-half a toothpick to make each "bond." The bonds are how the atoms are stuck together to make molecules.īuild each of the greenhouse gas molecules, as shown above. Here are all the greenhouse gas molecules, their formulas, and a picture of its gumdrop model. Study the gaseous elements and compounds in the Earth’s atmosphere nitrogen, ammonia, hydrogen, oxygen, metal and non-metal oxides and carbon dioxide. For example, ozone is also called O 3, where O stands for an oxygen atom and the little 3 means there are three of them. Each molecule has a shorthand name, which also gives its recipe, or formula. Gummy Greenhouse Gas models of these gases. Thereby, relating the amount of the substance to its constituent atoms or molecules. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms. Browse Getty Images premium collection of high-quality, authentic Carbon Dioxide Molecule stock photos, royalty-free images, and pictures. Hence, its molecular formula is given as $C$. The chemical formula for an ethylene monomer is -(CH2-CH2). One molecule of carbon-dioxide has a carbon atom covalently bonded to the two adjacent oxygen atoms. The substance may be atoms, ions or molecules. Hint: The mass of the substance is obtained by the sum of its constituent atoms and it is equal to its molecular weight in grams per mole of the substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
